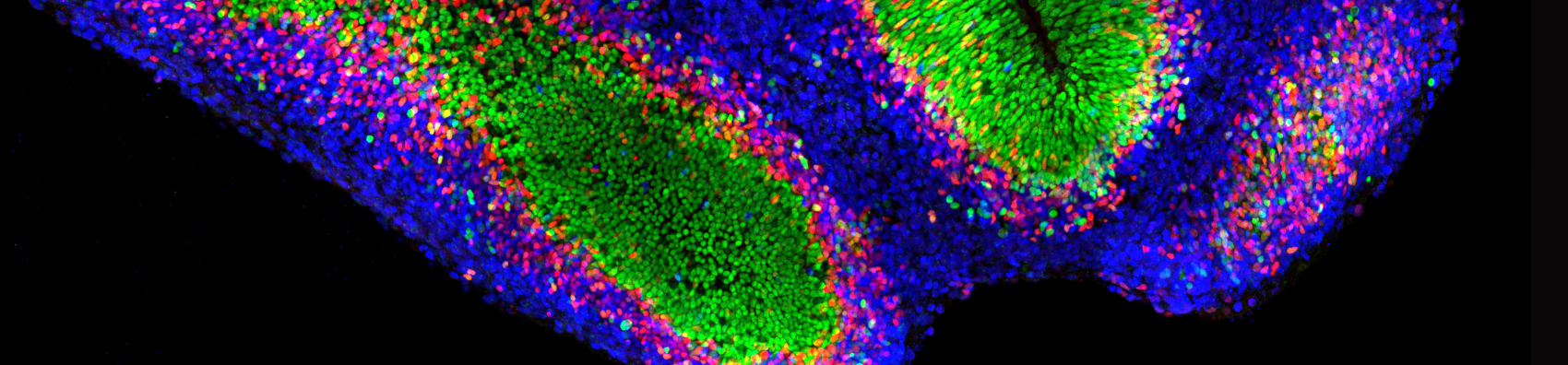
Program presentation
Cellular identities and destinies

A major public health issue
In France, pediatric brain tumors are the second most common cancer after leukemia in children under 15, with nearly 500 new cases diagnosed each year (2021 figures). Most patients who survive undergo intensive treatments, often resulting in long-term sequelae that impact their quality of life and integration into society. As such, managing these diseases poses a significant burden on individuals, families, and society as a whole.
In general, tumors are characterized by uncontrolled cell proliferation. The specific type of cancer depends on various factors, such as the nature of cellular deregulation, the type of cells involved, and the stage of life at which these deregulations occur.
What is a cell?
A single cell, the zygote (or egg), results from the fertilization of the oocyte by the sperm and enables the development of an entire organism. By dividing, it produces all the cells that make up the human body – over 30,000 billion distinct cells! These cells differ in their role yet share the same genetic information in their DNA. Every human being has a DNA, organized into 23 pairs of chromosomes, which codes for thousands of genes.
Trajectories that go off the rails
During an individual’s development, this genetic information can be read differently to produce different cell types such as neurons or muscle cells. In this way, cell trajectories or fates are adopted using different combinations of genes. These trajectories describe how a cell specializes and assumes a specific role in the organism. However, this reading of genetic information can be disrupted. In this case, the cell may follow an abnormal trajectory at the wrong time. This type of error can ultimately manifest itself in a variety of pathologies, including inflammatory and neurodegenerative diseases, as well as various types of cancer.
The Cell-ID program explores crucial questions in developmental biology, with a particular focus on neurodevelopment and pediatric brain cancers resulting from errors in the developmental program:
- How do cells acquire their identity? Every cell has a dynamically functioning genome, making it essential to understand how cell trajectories are generated over time
- How do diverse cell populations contribute to the three-dimensional organization of brain tissue? How each cell interacts in time and space with its neighbors and environment is an important issue in the formation of healthy tissue. Characterizing this blueprint, along with the distinct cell populations required for neurological development, remains a key challenge.
- How non-physiological deviations contribute to the onset and progression of brain tumors in children? Currently, children with brain tumors are often diagnosed at an advanced stage, when tumor cells have already spread extensively throughout the central nervous system. Advancements are needed to provide earlier, non-invasive diagnostic methods and detect the disease before disabling symptoms appear, as well as to monitor patients at risk and prevent relapses.
Three main obstacles to overcome
- The technologies needed to achieve cellular, spatial and time-dependent resolution in complex tissues are only now emerging, necessitating further development and refinement.
- The cost of these new technologies remains high, requiring specialized infrastructures, limiting their widespread implementation.
- The expertise required to implement and combine these new technologies calls for multidisciplinary research and a specific framework for cooperation and coordination between research institutes and hospital complexes.


Cell-ID’s mission
The Cell-ID research program “Cell identities and fates” aims to understand the mechanisms that govern cell identities and fates, and to determine when and how cells go off the rails. This involves studying the physiological development of the nervous system, and in parallel, certain pediatric brain cancers, whose origin is thought to be due to a deregulation of cell fate during embryonic development.
The approaches
This involves the use of imaging and functional genomics methods based in part on very high-throughput “next-generation” sequencing technologies, combined with the analysis and modeling of these data. We also need systems that mimic the properties of complex normal or pathological tissues. These combinations of approaches and tools are important assets for understanding when and how a cell follows a particular destiny under physiological conditions, in order to establish a benchmark. The same strategy can be applied to understand how cells lose their identity and deviate from their original fate during pathological conditions.
Expected results
Cell-ID, building on the LifeTime 2020 initiative and the concept of cellular interception medicine, has the potential to advance understanding of the cellular mechanisms underlying neurodevelopment, promote technological advances and foster the training of the next generation of interdisciplinary scientists. Ultimately, this initiative will pave the way for improved diagnosis, assessment of the risk of disease progression or recurrence, and adaptation of treatments as early as possible. The case of pediatric brain cancers represents a first example of pathology, but the same approaches could be applied to other pathologies in the future.
Four synergistic axes
- The targeted project (PC1) named CELL CONTEXT aims to identify optimal strategies combining emerging technologies, while developing new technologies to analyze over time and in cellular tissue how the genome functions at the scale of each cell.
- A second targeted project (PC2) named CELL EXP is dedicated to the development of model system approaches for the study of cell-cell interaction and the spatio-temporal monitoring of their fate. Researchers will use regionalized organoids – 3D multi-cell structures grown in vitro, mimicking specific regions of organs or tissues capable of simulating pathologies – as well as data visualization methods. These organoids will enable molecular tools to intervene on cell trajectories during embryonic development of the nervous system.
- The third targeted project (PC3), named DATA MED, integrates two main components. The first focuses on the development of predictive models for cell trajectories, utilizing existing biological data. These models will serve as a foundation for comparison with the second component, which involves generating new data from experimental models and tumor-derived samples. By combining these approaches, the project aims to derive initial hypotheses will be then experimentally tested using the most relevant tumor models.
- A fourth targeted project (PC4), CELL NEXT, aims to implement an original, tailor-made training and career development program. It will serve as a cement for inter-laboratory collaborations and innovative projects involving the entire Cell-ID community.
Key actions
- Recruitment of doctoral and post-doctoral students;
- Training and events through multidisciplinary symposia and summer schools;
- Communication campaigns aimed at the scientific community and the general public;
- Interactions with patient associations
- Setting up projects with a leverage effect ;
- Innovative projects for proof of concept ideas.
The discoveries resulting from this program will lead to major advancements in technologies, methodologies, and their application to the brain and selected pathologies. Beyond these pathologies, the progress achieved could extend to the treatment of other diseases. This project presents a unique opportunity to structure the French scientific community and position it at the forefront of international research in the promising field of cellular interception medicine.
